Project - Mapping trajectories of neurodegeneration in dementia
Research Objectives
- Our research program aims to build capacity for patient-tailored precision medicine in dementia by developing diagnostic and prognostic models of the pathophysiology of disease in-vivo. The research objectives are:
- To identify biological imaging markers of dementia subtypes;
- To predict their progression and determine their pathophysiology;
To enable clinical translation and patient stratification in disease-modifying interventions.
Project tag with a disease
Dementia
Research Project Description
The research program aims to construct a comprehensive picture of brain atrophy in dementia by separating phenotypic and temporal heterogeneity. Our research is carried out through ready-access to longitudinal cohorts across different syndromes and convergent methodologies (i.e., clinical phenotyping, multimodal neuroimaging, neuropathology, genetics and bioinformatics). The program includes 3 independent but complementary arms:
- Multimodal characterisation of brain atrophy. This arm develops multimodal imaging investigations using established neuroimaging methods (Fig 1) and novel bioinformatics approaches to obtain outcome measures of brain structure and function (Fig 2).
- Staging and prediction of trajectories of atrophy. This arm uses longitudinal modelling and neuroimaging biomarkers to predict disease trajectories (Fig 3).
- Patient-tailored precision medicine in dementia. This arm incorporates confirmed pathology post-mortem and discovery/prospective validation cohorts to obtain models of pathology subtype in-vivo.
Challenges within the field
Dementias are fatal and progressive neurodegenerative disorders. The key challenge for researchers and clinicians is heterogeneity. Even within clinical syndromes, individual patients experience significant variability in clinical presentation and underlying pathology. As such, accurate diagnosis of dementia in life is poor and only certain post-mortem. Disease duration is highly variable due to differing trajectories of disease progression in terms of the speed of brain atrophy and declining cognitive function. Thus, accurate prognosis and disease staging of dementia is currently not possible, as patients with similar presentations can face very different long-term outcomes. Critically, no cure for dementia currently exists. Disease-modifying treatments must engage biologically defined targets, and therefore a characterisation of dementias based on sensitive and specific biological markers (biomarkers) is needed.
People working on this project
- Dr Ramon Landin-Romero (Lead Investigator)
- Prof Olivier Piguet (Lead Investigator)
- Prof Glenda Halliday (Lead Investigator)
- Prof Fernando Calamante (Researcher, Collaborator)
- Prof Kirrie Ballard (Clinician, Collaborator)
- Dr Fiona Kumfor (Researcher, Collaborator)
- Dr Cristian Leyton (Researcher, Collaborator)
- Dr Yuichi Higashiyama (Researcher, International Collaborator)
- Prof John Kwok (Researcher, Collaborator)
- Dr Zac Chatterton (Researcher, Collaborator)
- A/Prof Rebekah Ahmed (Clinician, Collaborator)
- A/Prof Yasser Iturria-Medina (Researcher, International collaborator)
- Dr Yu (Sherry) Chen (Researcher, PhD Alumni, International collaborator)
- Mr Cheng Tao Liang (PhD Student)
- Mr James Carrick (Research Assistant)
Key Publications
- Leyton C, Landin-Romero R, Liang CT, Burrell J, Kumfor F, Hodges JR, Piguet O (2019). Correlates of Anomia in non-semantic Variants of Primary Progressive Aphasia Converge over Time. Cortex
- Ahmed RM, Landin-Romero R, Liang CT, Keogh JM, Henning E, Stickwerda-Brown C, Devenney E, Hodges JR, Kiernan MC, Farooqi IS & Piguet O (2019). Neural correlates of changes in body composition in frontotemporal dementia. Annals Clin Trans Neur
- Donnelly-Kehoe PA, Pascariello GO, García AM, Miller B, Rosen H, Manes F, Landin-Romero R, Matallana D, Serrano C, Herrera E, Reyes P, Santamaria-Garcia H, Kumfor F, Piguet O, Ibanez A, Sedeño L (2019). Robust automated computational approach for classifying frontotemporal neurodegeneration: a multimodal and multicentric neuroimaging study. Alzh & Dem DADM
- Chen Y, Kumfor F, Landin-Romero R, Irish, Hodges JR, Piguet O. The cerebellum in frontotemporal dementia: a meta-analysis of neuroimaging studies (2019). Neuropsych Reviews
- Chen Y, Kumfor F, Landin-Romero R, Irish, Hodges JR, Piguet O. Cerebellar atrophy and its contribution to cognitive dysfunction in the frontotemporal dementias (2018). Annals Neurology.
- McDonald S, Dalton KI, Rushby, JA & Landin-Romero R. Loss of white matter connectivity after severe traumatic brain injury (TBI) and its relationship to social cognition (2018). Brain Imaging and Behaviour (IF = 3.985)
- Landin-Romero, R. & Piguet O (2018). Recent advances in structural longitudinal neuroimaging of younger onset dementia. Neurodegener Dis Manag
- Midorikawa A, Kumfor F, Leyton, Foxe D, Landin-Romero, R & Piguet O. Characterization of positive behavioural changes in primary progressive aphasia (2018). Dem Geriatr Dis. (IF = 3.408)
- Landin-Romero, R., Kumfor, F., Leyton, C. E., Irish, M., Hodges, J. R., & Piguet, O. (2017). Disease-specific patterns of cortical and subcortical degeneration in a longitudinal study of Alzheimer's disease and behavioural-variant frontotemporal dementia. Neuroimage, 151, 72-80.
- Irish, M.†, Landin-Romero, R.†, Mothakunnel, A., Ramanan, S., Hsieh, S., Hodges, J. R., & Piguet, O. (2017). Evolution of autobiographical memory impairments in Alzheimer's disease and frontotemporal dementia - A longitudinal neuroimaging study. Neuropsychologia.
- O’Connor C, Landin-Romero R, Clemson L, Kaizik C, Daveson N, Hodges JR, Hsieh S, Piguet O & Mioshi E. (2017). Distinct functional profiles within behavioural variant frontotemporal dementia reflect different neuropsychiatric profiles: an imaging study. Neurology.
- Ahmed, R. M., Landin-Romero, R., Collet, T. H., van der Klaauw, A. A., Devenney, E., Henning, E., Kiernan, M. C., Piguet, O., Farooqi, I. S., & Hodges, J. R. (2017). Energy expenditure in frontotemporal dementia: a behavioural and imaging study. Brain, 140(Pt 1), 171-183
- Devenney, E. M., Landin-Romero, R., Irish, M., Hornberger, M., Mioshi, E., Halliday, G. M., Kiernan, M. C., & Hodges, J. R. (2017). The neural correlates and clinical characteristics of psychosis in the frontotemporal dementia continuum and the C9orf72 expansion.
- Sedeno, L., Piguet, O., Abrevaya, S., Desmaras, H., Garcia-Cordero, I., Baez, S., Alethia de la Fuente, L., Reyes, P., Tu, S., Moguilner, S., Lori, N., Landin-Romero, R., Matallana, D., Slachevsky, A., Torralva, T., Chialvo, D., Kumfor, F., Garcia, A. M., Manes, F., Hodges, J. R., & Ibanez, A. (2017). Tackling variability: A multicenter study to provide a gold-standard network approach for frontotemporal dementia. Hum Brain Mapp.
- Landin-Romero, R., Tan, R., Hodges, J. R., & Kumfor, F. (2016). An update on semantic dementia: genetics, imaging, and pathology. Alzheimers Res Ther, 8(1), 52
- Kumfor, F., Landin-Romero, R., Devenney, E., Hutchings, R., Grasso, R., Hodges, J. R., & Piguet, O. (2016). On the right side? A longitudinal study of left- versus right-lateralized semantic dementia. Brain, 139(Pt 3), 986-998. (IF = 10.103)
- Midorikawa A, Leyton CE, Foxe D, Landin-Romero R, Hodges JR, Piguet O. All Is Not Lost: Positive Behaviors in Alzheimer's Disease and Behavioral-Variant Frontotemporal Dementia with Disease Severity. J Alzheimers Dis. 2016 Sep 6;54(2):549-58.
Infographic / Medical Diagram / Scientific Diagram / Picture
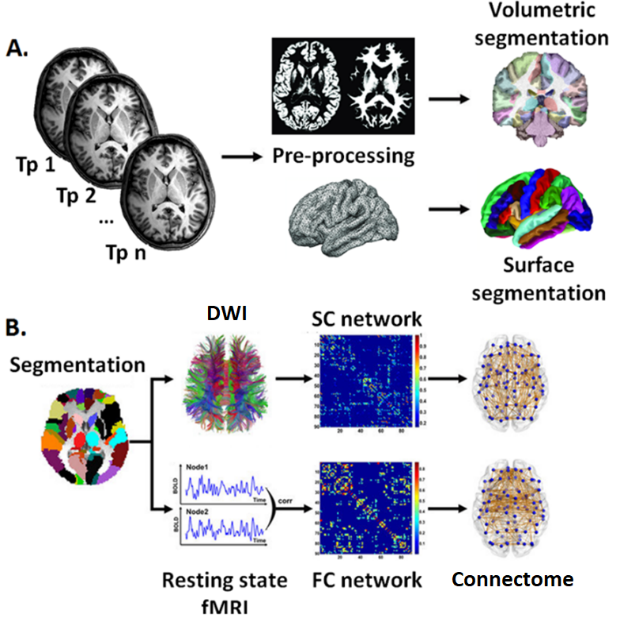
Figure 1. Schematic of multimodal analyses to obtain brain imaging outcome measures. (A) Structural stream; (B) Functional stream; Abbreviations: tp = time-point; DWI = Diffusion Weighted Imaging; SC = Structural connectivity; FC = Functional connectivity.
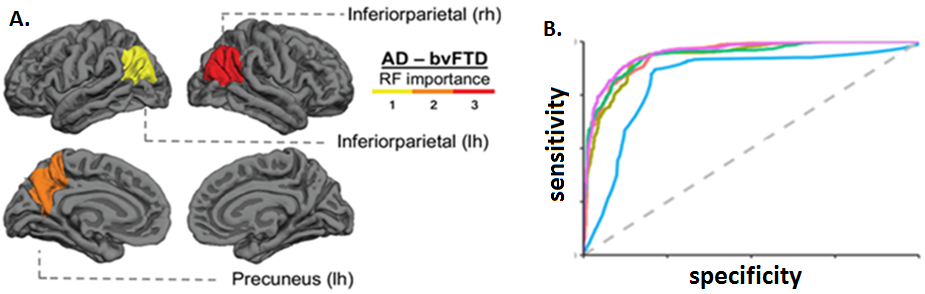
Figure 2. Pilot machine learning results of the diagnostic classifier. (A) Regions showing greater changes in AD vs behavioural-variant FTD (B) Inferioparietal atrophy (red colour, panel A) separates AD from FTD with 90% accuracy across ML classification algorithms (colour curves, panel B). Abbreviations: rh=right hemisphere; lh=left hemisphere; RF= relative feature importance
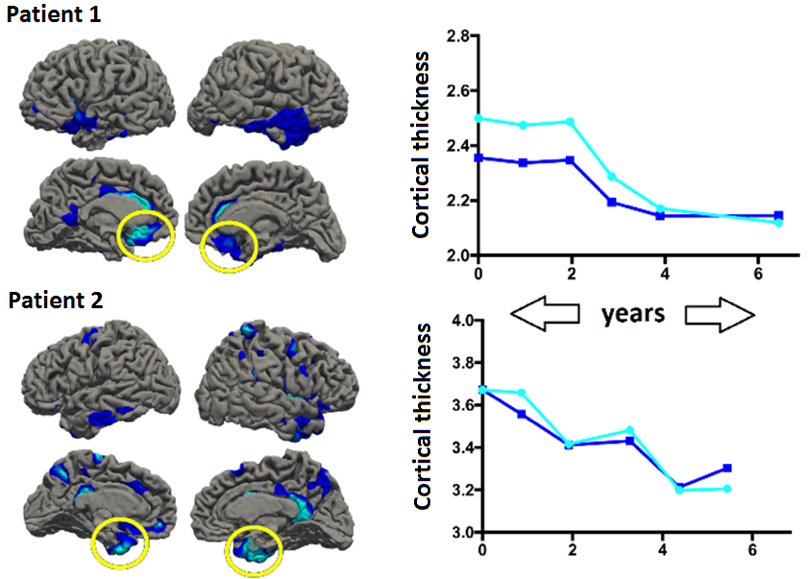
Figure 3. Pilot results of annualised, patient-level imaging trajectories. Patients 1 and 2 received the same diagnosis (FTD) but show different spread/rate of cortical thinning over time. Light/dark blue lines represent different epicentres of atrophy.
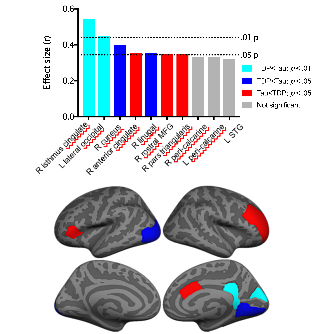
Figure 4. Differential patterns of progressive brain atrophy in the main pathology subtypes of frontotemporal dementia
Key Findings
- Our neuroimaging research has uncovered disease mechanisms of cognitive, language, emotional and metabolic dysregulation in young-onset dementia syndromes. We have progressed knowledge of the neural basis of disease trajectories in dementia, characterising common and syndrome-specific longitudinal brain changes, and contributed to the development of new neuroimaging methods.
- There are three outstanding challenges dementia; accurate diagnosis, prognosis and the urgent need of effective treatments. Our program addresses these challenges, which will be pivotal to our capacity to tackle dementia effectively.



